Vaccine manufacturing remains highly concentrated, despite new pandemic entrants

While new technologies such as mRNA have the promise to revolutionize vaccine manufacturing, they have not yet had an impact given that, with the exception of COVID-19 vaccines, candidates are still in development and manufacturing facilities have not yet come online, notes the World Health Organization (WHO) in its Global Vaccine Market Report 2022.
Ten new manufacturers
In 2021, based on data reported, 94 manufacturers supplied vaccines to WHO Member States. Ten of these entered the market in response to the COVID-19 pandemic (the entities include those performing all the steps of the vaccine manufacturing process, from producing drug substance through to final packaging, as well as fill/finish).
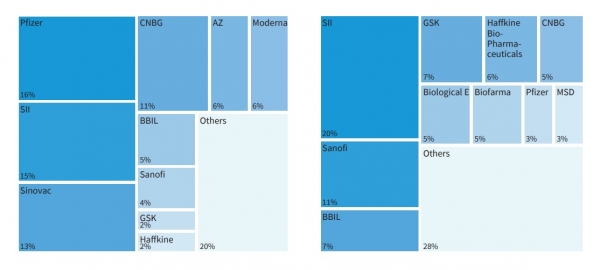
In 2021, COVID-19 vaccines ‘significantly changed’ the vaccine supply landscape. Various manufacturers emerged to capture global market value and volumes, with the top 10 manufacturers capturing almost 90% of the global value, indicating increased market concentration compared to all other vaccines.
And yet, excluding COVID-19 vaccines, the manufacturing landscape remains largely unchanged from 2019 in terms of the top manufacturing companies.
“Excluding COVID-19 vaccines, the manufacturers that are capturing the largest share of the global market value and supplying the majority of volume are largely unchanged, with 10 manufacturers supply 71% of the global volume," notes the report.
"The Serum Institute of India (SII), the China National Biotechnology Group (CNBG), Sanofi, GSK, MSD and Pfizer are the only manufacturers in the top 10 for both value and volume when excluding COVID-19 vaccines, similar to 2019. The global vaccine market therefore continues to be highly concentrated and reliant on a few manufacturers despite the new entrants during the COVID-19 pandemic.
“COVID-19 vaccines distributed in 2021 came from 19 manufacturers, 10 (52%) of which have no other marketed vaccine, and which collectively supplied 15% of the volume of COVID-19 vaccines. Only four COVID-19 vaccine manufacturers are in the top 10 in non-COVID-19 vaccine volumes, indicating that a significant portion of manufacturers of COVID-19 vaccines had limited prior experience in producing large volumes of vaccines.”

Problem areas: cholera, smallpox, ebola
The limited vaccine manufacturing base continues to be a cause for concern for the WHO.
“According to country data, globally, 10 manufacturers alone provide 70% of vaccine doses (excluding COVID-19 vaccines) and 85% of the global value of vaccines," notes the report.
"More importantly, when looking at individual vaccines, often only two or three suppliers provide more than 80% of supply. More than half of suppliers globally produce only a few vaccines and only serve local markets. Concentration of manufacturing has clear causes: vaccine development and manufacturing have high costs of entry and normally yield lower profits relative to the entry costs and compared to other pharmaceutical products.”
“In 2021, approximately 16 billion doses of 47 different vaccines, with an estimated value of $141bn, were distributed worldwide. This almost three-fold increase over the market volume in 2019 and 3.5-fold increase in market value was primarily driven by the advent of COVID-19 vaccines. Nevertheless, vaccines continue to represent a mere 4% of the overall pharmaceutical market (10% if we account for COVID-19 vaccines)."
Supply concentration leads to concerns on market health for key high-impact vaccines. The WHO outlines eight vaccines that could be needed to respond to public health emergencies: cholera, typhoid conjugate vaccine, Japanese encephalitis, MenA conj., MenAC conj. MenACYW-135 conj, smallpox/monkeypox and ebola.
Of these, six are considered to have ‘poor market health’ based on the risk of unpredictable surges in demand, low number of manufacturers or low commercial value (cholera, TCV, MenA, MenAC, smallpox/monkeypox and ebola), while only four are stockpiled (cholera, MenA conj, smallpox/monkeypox and ebola).
“The health of markets for six vaccines potentially needed in the event of an emergency, demand for which may surge during a disease outbreak, is also concerning. In addition, the manufacturing base is only in certain geographical areas: beyond China and India, vaccine manufacturers are mostly located in the EU, Indonesia, Japan and the USA. Lack of production capacity in various regions raises regional supply security concerns much discussed over the past decade and during the COVID-19 crisis: the African and Eastern Mediterranean Regions remain dependent on manufacturers headquartered elsewhere for 90% of their procured vaccines.”
Furthermore, each of the human papillomavirus, pneumococcal conjugate and measles, mumps and rubella combination vaccines is used by at least 100 countries, but each market is highly dependent on one or two manufacturers that account for more than 80% of vaccines by volume.
Promise of new tech
COVID-19 has shown that the production of vaccines can be scaled-up with new tech: but they have not yet been able to deliver an industry-wide shift.
“Among COVID-19 vaccines used in 2021, 36% were designed using innovative technologies and accounted for 59% of global volumes of COVID-19 vaccine procured by WHO Member States. Those innovative technologies have a large potential but are still in their infancy, as evidenced by their very limited use other than for COVID-19 vaccines.”
However, mRNA facilities from BioNTech and Moderna, for example, are expected to start coming online in the coming years: expected to offer not only COVID-19 vaccines but other vaccines if approved by regulatory authorities.
Public funding
Large public contracts to build vaccines manufacturing capacity and guarantee of purchase made a significant difference in the pandemic. The WHO, consequently, calls on governments to strengthen market preparedness by investing in new vaccine technologies, regional research and development and manufacturing hubs, as well as working towards regulatory harmonization.
“The vaccine market has a number of characteristics that render it highly complex and, thus, less attractive," notes the report. "It requires advanced know-how and is protected by intellectual property rights. Expansion of capacity implies non-linear production costs. Demand is very uncertain due to numerous factors, including competing health and non-health priorities, government fiscal space and prices.
“Without incentives, companies would have limited interest in investing in large manufacturing plants. Unprecedented use of partnerships has enhanced manufacturing bulk production, access to proprietary adjuvants and final product fill and finish. We should acknowledge the efforts that led manufacturers to make more than 10 billion doses of COVID-19 vaccine available by the end of 2021, while acknowledging that even more could have been done to meet larger global public health needs and support ambitious timelines for equitable vaccine deployment set by countries. Mainstreaming those successes and learnings outside of the unique circumstances of a pandemic will be necessary to address several of the shortcomings emerging in the vaccine ecosystem.”















