Merck aiming to help biopharmas make CHO cells resistant to MVM

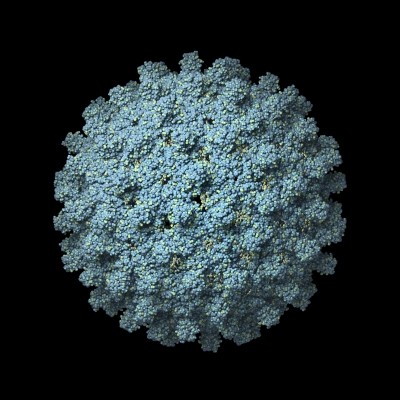
The virus in question is MVM (minute virus of mice). It infects and kills rodents and cells derived from such species – including Chinese hamster ovary (CHO) cells - but has no impact on humans.
MVM infects CHO cells by binding 2,3-linked sialic acid residues on their outer membranes. This triggers the virus to be taken into the cell by other proteins.
It would be possible to prevent infection by knocking out the genes that encode the sialic acid receptors, however these proteins also help determine the glycosylation profiles of proteins the CHO cells express.
Any alteration of these genes would therefore have a detrimental impact on biopharmaceuticals produced in the modified cells.
A new service – called the Centinel programme – aims to prevent MVM infection using another approach.
The approach uses the zinc finger nuclease (ZFN) technology - which Germany-based Merck gained when it bought Sigma-Aldrich in 2014 - to edit the genes that encode proteins involved in viral uptake rather than binding.
MVM infection
MVM has been a problem for a number of biopharmaceutical firms, notably Roche’s Genentech unit which lost product on several occasions as a result of MVM contamination in the early 1990s.
Genentech’s response was to begin heat treating the serum it uses to grow CHO cells, a task which also involved proving to agencies like the US Food and Drug Administration (FDA) and European Medicines Agency (EMA) that the process did not alter the protein being made.
Regulators around the world require that biopharmaceutical firms test CHO cells for viral contamination. Such processes are technically challenging, time consuming and expensive.
Service
Merck did not respond to a request for more information about the Centinel programme.
However, in a press statement the firm said its Bioreliance division – also acquired in 2014 – will “validate MVM resistance and demonstrate the virus is not propagated in the cell line.”