Animal study shows benefit from gene therapy in late-stage eye disease

This is the significance of a new study that successfully preserved the vision of dogs suffering from naturally occurring, late-stage retinitis pigmentosa. Up to now, animal studies had shown benefits from gene therapy only when it was used in the earliest stages of the disease.
This research succeeded in improving retinal cells and preventing vision loss for 2 years in man’s best friend with a common and severe form of this eye disease.
“This is the first study to show that the disease can already be seen and yet be corrected and that no further degeneration will happen,” Neeraj Agarwal, program director at the National Eye Institute which funded the research, told Biopharma-Reporter.com.
The research “provides good proof of concept for gene therapy at the intermediate stage of the disease, thus widening the therapeutic window,” he explained.
Mutated RPGR genes
Retinitis pigmentosa is the most common inherited disease that causes degeneration of the retina, light-sensitive tissue in the eye. Around 1 in 4,000 people are impacted, with 10 to 20% having a severe form called X-linked retinitis pigmentosa. This mainly affects males, causing night blindness and progressive loss of vision.
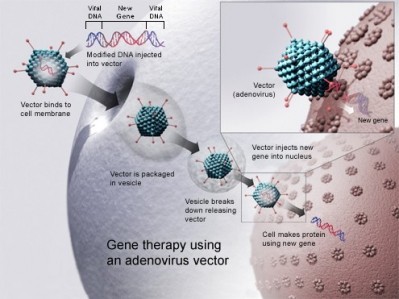
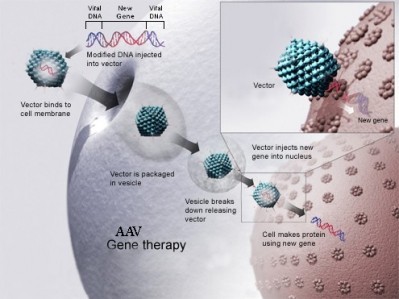
The cause is mutated RPGR (retinitis pigmentosa GTPase regulator) genes, important for healthy photoreceptors. The researchers tested the gene therapy in a canine model of this condition, reporting the results in the Proceedings of the National Academy of Sciences.
“Viruses carried the normal RPGR gene to rod and cone photoreceptor cells in a region of the retina. In that treated region the progressive loss of photoreceptors was arrested whereas in surrounding untreated regions the loss of photoreceptors continued,” explained co-author Artur Cideciyan, University of Pennsylvania Perelman School of Medicine, Philadelphia.
Human trials?
Imaging confirmed that the human RPGR transgene was stably expressed in photoreceptors and linked to improvements in rods, cones and other retinal cells.
Gene therapy stopped disease-associated cell death for at least the 2.5 year length of the study, even in dogs with later-stage disease. Additional research is now needed to assess the therapy’s safety and the risk for potential toxicity in patients.
“We are hoping that in 2 or 3 years we will receive a proposal [from some of the authors] to fund a human trial, so that in perhaps 5 years we can show the feasibility of this approach in a few patients,” says Agarwal. Thereafter phase I trials may begin.