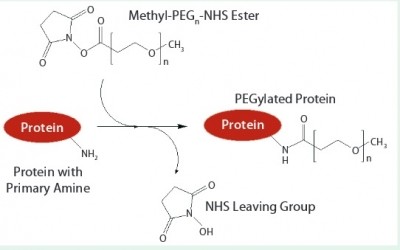
Cobra works with QuiaPEG on PEGylation for extended half-life

Cobra’s Commercial Director Julian Hanak told BioPharma-Reporter.com client demand is growing for PEGylated large molecules – proteins made bigger by attaching a large inert polymer (polyethylene glycol, or PEG) to extend their stay in the body.
“The reason you do that is if a protein is very small it is excreted by your kidneys. So once a patient is dosed with the protein it doesn’t stay in the body very long. By making the protein bigger by attaching a large intert polymer, it survives in the body for longer.
“It’s very similar to the antifreeze you put in your car radiator –that’s also a form of PEGylation.”
Lower dose, higher compliance
PEGylation has several advantages for biopharmaceuticals, Hanak told us.
“The patient gets dosed with less of the product – so we see fewer side effects, and a decreased cost of goods. Patient compliance is much improved. For some versions of proteins, the un-PEGylated form has to be taken once a day – but these drugs can be taken once a week or even once a month. So patients experience better control of their medicine.
“One of the reasons why insulin patients suffer is the concentration of the drug goes up and down. Patients are very non-compliant with dosing themselves – they don’t like it or it makes them feel ill. So they spend more time outside the therapeutic window.”
Biobetter opportunities
Hanak said Cobra had chosen Swedish biotech company QuiaPEG for this alliance because its platform is “a second-generation PEGylation technology that offers significant improvements on the existing technology.
“With the existing technologies, when you modify your protein with PEGylation, it has two reactive domains at either end of the molecule. A significant proportion of the product you make is cross-linked – you don’t want that. The QuiaPEG technology is mono-functional – it therefore produces much more of the correct product, using less of the drug.”
Hanak said client demand for PEGylated production is driven in part by second-generation antibody products: “The success of monoclonal antibodies means a lot of pharmaceutical companies are looking at making antibody fragments. These are very small, so half-life extension is requirement for those.”
Secondly, he said, life-cycle management was another driver. As biologics come off patent, “original companies are looking to extend their marketing exclusivity, and protein modification can be an answer.”
ADCs: ‘Work to do’
Looking ahead, Cobra’s expert told us “there’s still a lot of work to do” around the chemistry of PEGylation. “At the moment, where the PEG binds to the protein is generally a random event.
Companies are interested in “targeted conjugation,” he said, “so you have the PEG away from the active domain of the protein. The ideal product for us is a mono-dispersed product – where the legs of the polymer are very uniform.”
He added that PEGylation could be used on antibody drug conjugates (ADCs), improving their solubility, and reducing their toxicity.
“The major change is that more and more of our customers are no longer interested in naturally occurring molecules they want to replace to cure a disease. More commonly they have designed a protein of their own in silico [on a computer].
“These molecules are entirely synthetic– nature hasn’t learned how to build and express them. They can therefore be designed to target exactly what they want and tend to be smaller and more potent.”
However, Hanak explained, these proteins, while promising, “need to be made more soluble, and to reduce problems like immunogenicity, and aggregation,” a problem PEGylation may be able to solve.